The Problem
What Conditions cause the warming of the atmosphere?
Hypothesis
If two pans that are filled with water are place under a lamp, that represents the sun, and if one of those pans is covered with saran wrap, that represents the ozone layer, then the one with the saran wrap will take longer to heat up because the saran wrap will protect the water from the heat coming from the lamp.
Variables
Independent: Saran Wrap
Dependent: Change in Temperature
Controlled: amount of water in pan, size of pans, time under light
Dependent: Change in Temperature
Controlled: amount of water in pan, size of pans, time under light
Experimental Groups
Control Group: the pan without the saran wrap serving as an "ozone layer"
Experimental Group: the pan with the saran wrap serving as an "ozone layer"
Experimental Group: the pan with the saran wrap serving as an "ozone layer"
Materials
2 pans
water
heat lamp
thermometer
stopwatch
saran wrap
beaker
water
heat lamp
thermometer
stopwatch
saran wrap
beaker
Procedure
1. Fill one pan with 1000mL of water using a beaker to measure
2. Record the initial temperature of the water by putting a thermometer into the water for 30 seconds
3. Apply the heat lamp to the pan of water for 14 minutes
4. Record the temperature of the water every 2 minutes by sticking the thermometer into the water for 30 seconds
5. Dump the water
6. Fill the other identical pan iwth 1000mL of water using a beaker to measure
7. Record the initial temperature of the water by putting a thermometer into the water for 30 seconds
8. Apply saran wrap to the top of the pan
9. Apply the heat lamp to the pan of water for 14 minutes
10. Record the temperature of the water every 2 minutes by sticking the thermometer into the water for 30 seconds
11. Dump the water
12. Examine Data
2. Record the initial temperature of the water by putting a thermometer into the water for 30 seconds
3. Apply the heat lamp to the pan of water for 14 minutes
4. Record the temperature of the water every 2 minutes by sticking the thermometer into the water for 30 seconds
5. Dump the water
6. Fill the other identical pan iwth 1000mL of water using a beaker to measure
7. Record the initial temperature of the water by putting a thermometer into the water for 30 seconds
8. Apply saran wrap to the top of the pan
9. Apply the heat lamp to the pan of water for 14 minutes
10. Record the temperature of the water every 2 minutes by sticking the thermometer into the water for 30 seconds
11. Dump the water
12. Examine Data
Observations
Pan without saran wrap:
10 minutes: little bubbles forming on the bottom of the pan (like the start of a slow boil?)
Didn't get as extreme of results as expected b/c we didn't realize how long that amount of water takes to heat up
Pan with saran wrap:
Saran wrap is fogged up
14 minutes: it feels hot under the saran wrap like as if it's trapping heat
10 minutes: little bubbles forming on the bottom of the pan (like the start of a slow boil?)
Didn't get as extreme of results as expected b/c we didn't realize how long that amount of water takes to heat up
Pan with saran wrap:
Saran wrap is fogged up
14 minutes: it feels hot under the saran wrap like as if it's trapping heat
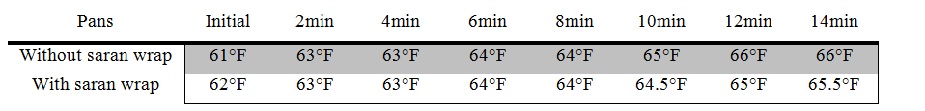
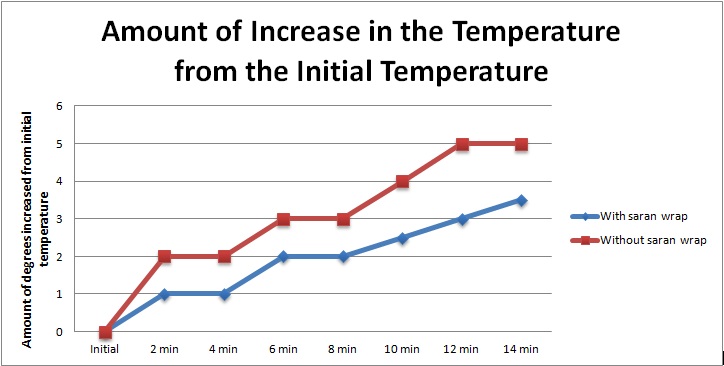
Data Tables and Graph
Analysis and Conclusion
1. Evaluate the Hypothesis:
The results did support my hypothesis. The temperature of the pan with the saran wrap ultimately increased by 3.5 degrees farenheit while the temperature of the pan without the saran wrap ultimately increased by 5 degrees farenheit; therefore, my hypothesis was proved to be correct.
2. Possible Sources of Error:
The experimental design did not go as planned. The pan with the saran wrap did heat up faster just like we thought it would; however, I don't think that we got very accurate readings because our initial temperatures were not the same and I think that we should have left the thermometer in the water and checked it every 2 minutes instead of putting it in every 2 minutes for 30 seconds. I also don't think that 30 seconds was a long enough time to get an accurate reading from the thermometer. Another thing that we could have changed to make the experiment more accurate would be to leave the water under the lamps for a longer period of time to see if the initial pattern stayed consistant.
3. Application
The experiment proved that water takes a long time for the temperature to change in general. The information of this experiment added to the world's knowledge that water does not heat up extremely quickly especially when covered with some sort of barrier. This information can be useful to scientists who are trying to figure out how to prevent the melting of glaciers. Although the matter of glacier melting should be taken seriously, the ozone may make the ice melt slower than we think. Since the sun does not heat up the earth's water immediately, the steady melting of glaciers may continue for many years until all the ice is melted.
The results did support my hypothesis. The temperature of the pan with the saran wrap ultimately increased by 3.5 degrees farenheit while the temperature of the pan without the saran wrap ultimately increased by 5 degrees farenheit; therefore, my hypothesis was proved to be correct.
2. Possible Sources of Error:
The experimental design did not go as planned. The pan with the saran wrap did heat up faster just like we thought it would; however, I don't think that we got very accurate readings because our initial temperatures were not the same and I think that we should have left the thermometer in the water and checked it every 2 minutes instead of putting it in every 2 minutes for 30 seconds. I also don't think that 30 seconds was a long enough time to get an accurate reading from the thermometer. Another thing that we could have changed to make the experiment more accurate would be to leave the water under the lamps for a longer period of time to see if the initial pattern stayed consistant.
3. Application
The experiment proved that water takes a long time for the temperature to change in general. The information of this experiment added to the world's knowledge that water does not heat up extremely quickly especially when covered with some sort of barrier. This information can be useful to scientists who are trying to figure out how to prevent the melting of glaciers. Although the matter of glacier melting should be taken seriously, the ozone may make the ice melt slower than we think. Since the sun does not heat up the earth's water immediately, the steady melting of glaciers may continue for many years until all the ice is melted.
Real Life Example
Watts, Susan. "Arctic Ice Melt 'like Adding 20 Years of CO2 Emissions'" BBC News. BBC, 09 June 2012. Web. 02 Feb. 2013.
http://www.bbc.co.uk/news/science-environment-19496674.
http://www.bbc.co.uk/news/science-environment-19496674.